26.11.2020 | The first COVID-19 vaccine is getting FDA emergency authorization
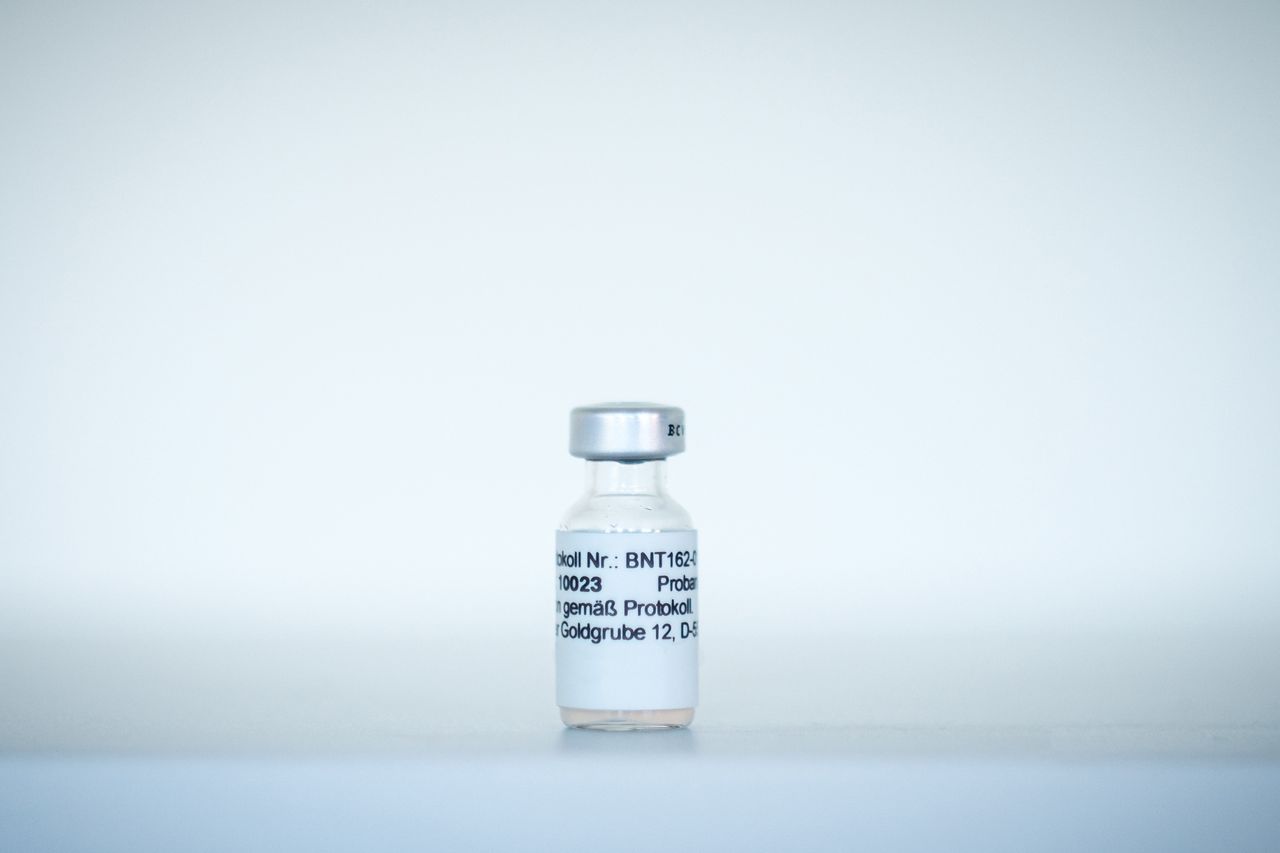
On Friday, last week, the US Food and Drug Administration have received their first submission of a Corona Virus Vaccine.
Pharmaceutical giant Pfizer and German biotech firm BioNTech have announced that they are submitting the formal request to obtain an Emergency Use Authorization (EUA) from the FDA for their mRNA vaccine, BNT162b2.
In the trial for the vaccine, only 170 cases occurred among the 43,661 participants, who were enrolled at one of 150 trial sites in the United States, Germany, Turkey, South Africa, Brazil, and Argentina. Of the 170 cases, 162 were in the placebo group and just eight occurred in the group receiving the experimental two-dose vaccine. Among all the cases, 10 were severe, nine of which were in the placebo group.















